Takeaways
- The greenhouse effect is essential to life on Earth, but human-made emissions in the atmosphere are trapping and slowing heat loss to space.
- Five key greenhouse gases are carbon dioxide, nitrous oxide, methane, chlorofluorocarbons, and water vapor.
- While the Sun has played a role in past climate changes, the evidence shows the current warming cannot be explained by the Sun.
Increasing Greenhouses Gases Are Warming the Planet
Scientists attribute the global warming trend observed since the mid-20th century to the human expansion of the "greenhouse effect"1 — warming that results when the atmosphere traps heat radiating from Earth toward space.
Life on Earth depends on energy coming from the Sun. About half the light energy reaching Earth's atmosphere passes through the air and clouds to the surface, where it is absorbed and radiated in the form of infrared heat. About 90% of this heat is then absorbed by greenhouse gases and re-radiated, slowing heat loss to space.
Four Major Gases That Contribute to the Greenhouse Effect
Carbon Dioxide
A vital component of the atmosphere, carbon dioxide (CO2) is released through natural processes (like volcanic eruptions) and through human activities, such as burning fossil fuels and deforestation.
Methane
Like many atmospheric gases, methane comes from both natural and human-caused sources. Methane comes from plant-matter breakdown in wetlands and is also released from landfills and rice farming. Livestock animals emit methane from their digestion and manure. Leaks from fossil fuel production and transportation are another major source of methane, and natural gas is 70% to 90% methane.
Nitrous Oxide
A potent greenhouse gas produced by farming practices, nitrous oxide is released during commercial and organic fertilizer production and use. Nitrous oxide also comes from burning fossil fuels and burning vegetation and has increased by 18% in the last 100 years.
Chlorofluorocarbons (CFCs)
These chemical compounds do not exist in nature – they are entirely of industrial origin. They were used as refrigerants, solvents (a substance that dissolves others), and spray can propellants.
FORCING: Something acting upon Earth's climate that causes a change in how energy flows through it (such as long-lasting, heat-trapping gases - also known as greenhouse gases). These gases slow outgoing heat in the atmosphere and cause the planet to warm.

Another Gas That Contributes to the Greenhouse Effect:
Water Vapor
Water vapor is the most abundant greenhouse gas, but because the warming ocean increases the amount of it in our atmosphere, it is not a direct cause of climate change.
Credit: John Fowler on UnsplashFEEDBACKS: A process where something is either amplified or reduced as time goes on, such as water vapor increasing as Earth warms leading to even more warming.

Human Activity Is the Cause of Increased Greenhouse Gas Concentrations
Over the last century, burning of fossil fuels like coal and oil has increased the concentration of atmospheric carbon dioxide (CO2). This increase happens because the coal or oil burning process combines carbon with oxygen in the air to make CO2. To a lesser extent, clearing of land for agriculture, industry, and other human activities has increased concentrations of greenhouse gases.
The industrial activities that our modern civilization depends upon have raised atmospheric carbon dioxide levels by nearly 50% since 17502. This increase is due to human activities, because scientists can see a distinctive isotopic fingerprint in the atmosphere.
In its Sixth Assessment Report, the Intergovernmental Panel on Climate Change, composed of scientific experts from countries all over the world, concluded that it is unequivocal that the increase of CO2, methane, and nitrous oxide in the atmosphere over the industrial era is the result of human activities and that human influence is the principal driver of many changes observed across the atmosphere, ocean, cryosphere and biosphere.
"Since systematic scientific assessments began in the 1970s, the influence of human activity on the warming of the climate system has evolved from theory to established fact."

Intergovernmental Panel on Climate Change
The panel's AR6 Working Group I (WGI) Summary for Policymakers report is online at https://www.ipcc.ch/report/ar6/wg1/.
Evidence Shows That Current Global Warming Cannot Be Explained by Solar Irradiance
Scientists use a metric called Total Solar Irradiance (TSI) to measure the changes in energy the Earth receives from the Sun. TSI incorporates the 11-year solar cycle and solar flares/storms from the Sun's surface.
Studies show that solar variability has played a role in past climate changes. For example, a decrease in solar activity coupled with increased volcanic activity helped trigger the Little Ice Age.
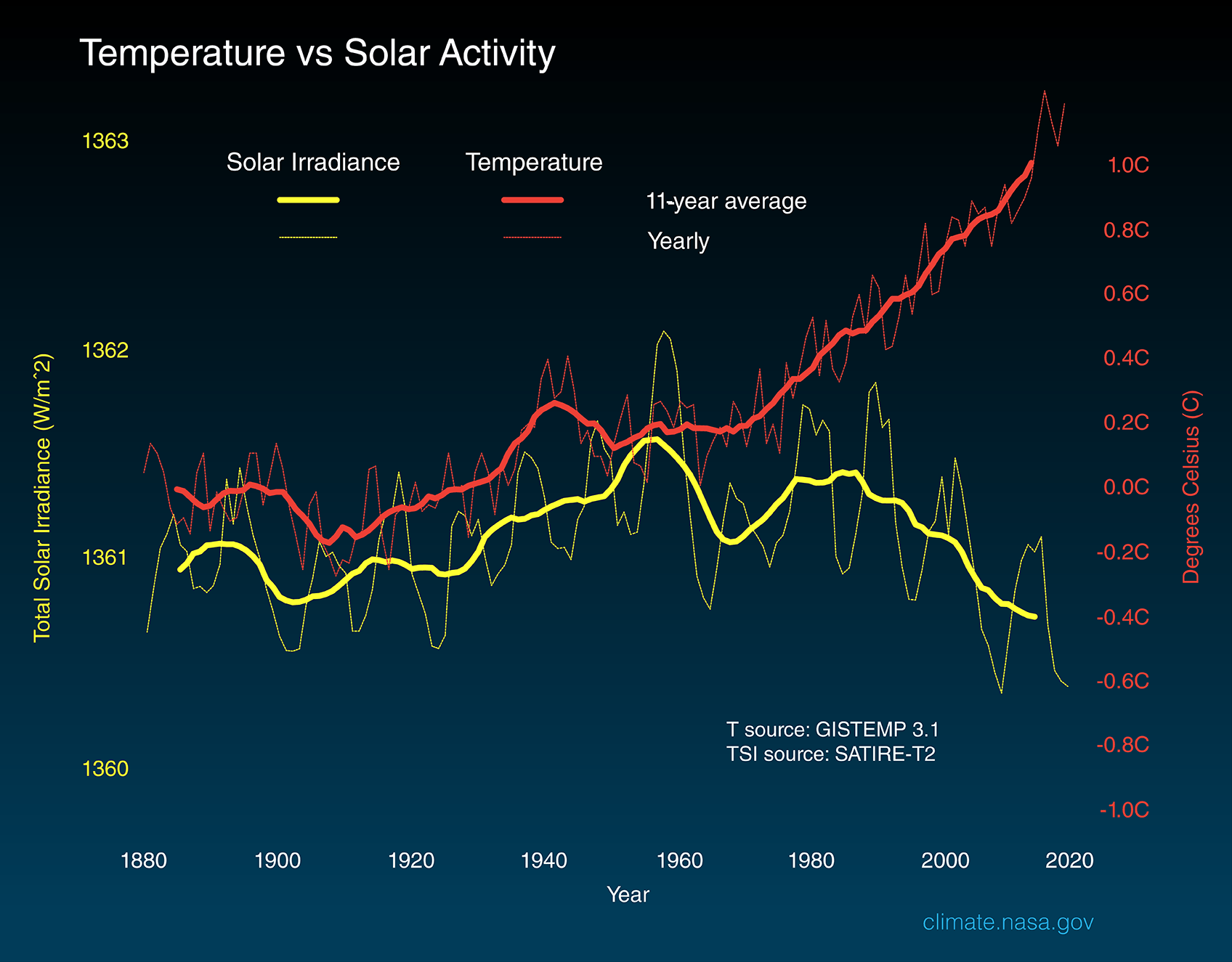
The amount of solar energy that Earth receives has followed the Sun’s natural 11-year cycle of small ups and downs with no net increase since 1880. Over the same period, global temperature has risen markedly. It is therefore extremely unlikely that the Sun has caused the observed global temperature warming trend over the past half-century.
But several lines of evidence show that current global warming cannot be explained by changes in energy from the Sun:
- Since 1750, the average amount of energy from the Sun either remained constant or decreased slightly3.
- If a more active Sun caused the warming, scientists would expect warmer temperatures in all layers of the atmosphere. Instead, they have observed a cooling in the upper atmosphere and a warming at the surface and lower parts of the atmosphere. That's because greenhouse gases are slowing heat loss from the lower atmosphere.
- Climate models that include solar irradiance changes can’t reproduce the observed temperature trend over the past century or more without including a rise in greenhouse gases.
References
1. IPCC 6th Assessment Report, WG1, Summary for Policy Makers, Sections A, “The Current State of the Climate”
IPCC 6th Assessment Report, WG1, Technical Summary, Sections TS.1.2, TS.2.1 and TS.3.1
2. P. Friedlingstein, et al., 2022: “Global Carbon Budget 2022”, Earth System Science Data (11 Nov 2022): 4811–4900. https://doi.org/10.5194/essd-14-4811-2022
3. IPCC 6th Assessment Report, WG1, Chapter 2, Section 2.2.1, “Solar and Orbital Forcing”
IPCC 6th Assessment Report, WG1, Chapter 7, Sections 7.3.4.4, 7.3.5.2, Figure 7.6, “Solar”
M. Lockwood and W.T. Ball, Placing limits on long-term variations in quiet-Sun irradiance and their contribution to total solar irradiance and solar radiative forcing of climate,” Proceedings of the Royal Society A, 476, issue 2228 (24 June 2020): https://doi 10.1098/rspa.2020.0077
Header image credit: Pixabay/stevepb
Four Major Gases image credit: Adobe Stock/Ilya Glovatskiy